Pharma Automation: Transforming Pharmaceutical Operations
Share on
The pharmaceutical industry faces unprecedented pressure to deliver products faster, maintain stringent quality standards, and comply with complex regulations across global markets. Manual processes that once served as the backbone of pharmaceutical operations now struggle to meet the demands of modern supply chains, where speed, accuracy, and traceability are non-negotiable. Pharma automation has emerged as the critical solution, combining advanced robotics, intelligent software systems, and seamless integration to transform how pharmaceutical companies manage their warehousing, manufacturing, and distribution operations.
Understanding Pharma Automation in Modern Supply Chains
Pharma automation encompasses the sophisticated technologies and systems that replace or enhance manual processes across pharmaceutical operations. This transformation extends far beyond simple mechanisation, incorporating artificial intelligence, machine learning, and advanced robotics to create intelligent, responsive environments that adapt to changing demands whilst maintaining the rigorous standards pharmaceutical operations require.
The scope of automation in pharmaceutical settings ranges from automated storage and retrieval systems (AS/RS) that manage inventory with pinpoint accuracy to robotic picking solutions that handle products with care and precision. These systems integrate with warehouse management software (WMS) and enterprise resource planning (ERP) platforms to create seamless workflows that track every product movement, temperature exposure, and handling event throughout the supply chain.
Key Components of Pharmaceutical Automation Systems
Modern pharmaceutical automation systems comprise several interconnected technologies working in concert:
- Robotic material handling for safe product movement and placement
- Automated storage systems optimised for temperature-controlled environments
- Vision systems for quality control and verification
- Track-and-trace technology ensuring complete product genealogy
- Environmental monitoring maintaining proper storage conditions
- Integration platforms connecting warehouse, manufacturing, and enterprise systems
These components don't operate in isolation. The real power of pharma automation emerges when these systems communicate seamlessly, creating an intelligent ecosystem that responds to demand fluctuations, prioritises urgent orders, and maintains compliance documentation automatically.


Addressing Pharmaceutical Industry Challenges Through Automation
The pharmaceutical sector confronts unique operational challenges that make automation not merely beneficial but essential for competitive survival. Product sensitivity, regulatory requirements, and supply chain complexity create an environment where manual processes introduce unacceptable risk.
Temperature excursions represent a critical concern for pharmaceutical products, many of which require specific storage conditions to maintain efficacy. Automated systems equipped with environmental sensors and control mechanisms maintain precise conditions whilst documenting every temperature reading, creating the audit trail regulatory bodies demand. When deviations occur, these systems trigger immediate alerts and can automatically quarantine affected inventory before human intervention.
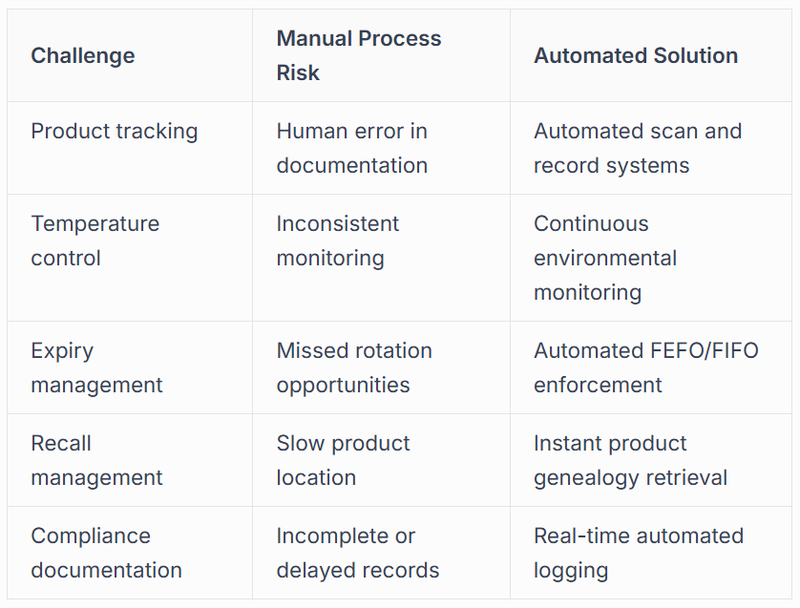

Serialisation requirements under regulations like the Drug Supply Chain Security Act (DSCSA) demand that pharmaceutical companies track products at the individual unit level. Manual serialisation processes prove labour-intensive and error-prone, whilst automation solutions for pharmaceutical operations handle serialisation, aggregation, and verification at speeds manual processes cannot approach.
Compliance and Regulatory Advantages
Pharmaceutical automation directly addresses the compliance burden that consumes significant resources in traditional operations. Automated systems create comprehensive documentation of every process step, from receiving through storage to picking and shipping. This documentation occurs in real-time, eliminating the delays and gaps that characterise manual record-keeping.
Good Distribution Practice (GDP) and Good Manufacturing Practice (GMP) requirements specify detailed protocols for handling pharmaceutical products. Automated systems can be programmed to follow these protocols precisely, executing them consistently without the variability inherent in human performance. When audits occur, automated systems provide complete, time-stamped records that demonstrate compliance far more convincingly than manual logs.
Warehouse Automation Solutions for Pharmaceutical Operations
Pharmaceutical warehouse environments demand specialised automation approaches that balance efficiency with the sector's unique requirements. Unlike general logistics operations, pharmaceutical warehousing must prioritise product integrity, environmental control, and regulatory compliance alongside traditional metrics like throughput and accuracy.
Goods-to-person (GTP) systems have revolutionised pharmaceutical warehouse operations by bringing products directly to operators in controlled workstations. These systems eliminate travel time, reduce physical strain on workers, and create ergonomic environments where operators can focus on accuracy rather than rushing through picks. For pharmaceutical operations managing thousands of SKUs with varying storage requirements, GTP systems offer the flexibility to segregate products by temperature zone, security level, or handling requirements whilst maintaining efficient workflows.
Starting a warehouse automation journey can feel daunting for pharmaceutical distributors managing growing volumes. The Automate-X GTP Starter Grid provides an accessible entry point, offering a scalable goods-to-person solution designed specifically for small and medium businesses in Australia and New Zealand. This approach allows pharmaceutical distributors to begin automating their picking processes without the massive capital investment traditionally associated with warehouse automation, whilst building the foundation for future expansion.


Temperature-Controlled Automation
Temperature control represents one of the most critical aspects of pharmaceutical warehousing, with products ranging from ambient to frozen storage requirements. Automated systems designed for pharmaceutical applications incorporate multiple temperature zones within a single facility, moving products between zones as needed whilst maintaining environmental integrity.
Modern pharma automation systems employ several strategies for temperature management:
- Segregated storage zones with dedicated environmental controls
- Automated environmental monitoring with continuous logging
- Insulated transfer mechanisms for inter-zone product movement
- Backup power systems ensuring uninterrupted climate control
- Alert protocols for immediate response to temperature deviations
These systems integrate with cold chain monitoring platforms, creating complete temperature histories for every product. When products move through distribution networks, this data travels with them, providing receiving facilities with confidence in product integrity and creating the documentation regulatory bodies require.


Manufacturing Support and Production Integration
Pharma automation extends beyond warehousing into manufacturing support operations where automation systems supply production lines, manage work-in-progress inventory, and handle finished goods. This integration creates seamless material flow from raw material receipt through production to finished product storage and distribution.
Automated guided vehicles (AGVs) and autonomous mobile robots (AMRs) transport materials between storage locations and production areas on programmed schedules or on-demand requests. These systems eliminate manual material handling, reduce contamination risks, and ensure production lines receive materials precisely when needed. Integration with manufacturing execution systems (MES) allows these robots to respond dynamically to production changes, adjusting delivery sequences and quantities automatically.
The connection between warehouse automation and manufacturing creates powerful opportunities for pharmaceutical operations. Production scheduling systems can query warehouse inventory in real-time, adjusting production plans based on material availability. Conversely, warehouse systems can prepare materials for production runs before they're requested, staging components near production areas to eliminate delays.
Quality Control and Inspection Automation
Quality control represents a critical checkpoint in pharmaceutical operations, where products undergo rigorous inspection before release. Advanced automation technologies integrate vision systems, weight verification, and serialisation readers into automated workflows, conducting multiple inspection steps simultaneously as products move through the system.
These automated inspection processes achieve consistency impossible with manual checks. Vision systems examine every unit for packaging defects, label accuracy, and physical damage at speeds measured in units per second rather than units per minute. Weight verification systems detect missing components or incorrect product configurations. Serialisation readers confirm that every unit carries the correct identification and that aggregation hierarchies match shipping documents.
When inspection systems detect anomalies, they can automatically divert affected products to quarantine locations whilst alerting quality personnel. This immediate response prevents defective products from reaching customers whilst creating detailed records of what was found, when, and how it was handled.
Robotics and Advanced Handling Technologies
The evolution of robotic automation in pharmaceutical environments has accelerated dramatically as robots become more sophisticated, flexible, and capable of handling the delicate products pharmaceutical operations require. Modern collaborative robots (cobots) work alongside human operators, handling repetitive tasks whilst allowing humans to focus on decision-making and exception handling.
Robotic systems excel at tasks requiring precise, repeatable movements performed thousands of times daily. In pharmaceutical applications, this includes:
- Unit picking for small-item orders requiring individual product selection
- Case handling for efficient pallet building and depalletisation
- Kitting operations assembling procedure packs or patient-specific orders
- Inspection assistance presenting products to cameras or operators for verification
- Packaging processes placing products into shipping containers with precise orientation
The pharmaceutical industry's exploration of unmanned warehouse concepts demonstrates how far automation capabilities have advanced. Fully automated facilities operate with minimal human intervention, handling receiving, storage, picking, and shipping operations through coordinated robotic systems that work continuously, maintaining consistent accuracy regardless of shift or season.
Material Handling for Sensitive Products
Pharmaceutical products often require specialised handling due to their fragility, sensitivity to environmental conditions, or regulatory requirements. Vacuum conveying solutions designed specifically for pharmaceutical applications transport powders and granular materials safely, preventing contamination whilst maintaining product integrity throughout the transfer process.
These systems address the challenges of moving sensitive materials between production stages, storage locations, and packaging lines. Enclosed transfer mechanisms prevent exposure to ambient conditions, protect products from contamination, and eliminate the dust generation that characterises manual handling of powdered materials. Integration with process control systems allows these conveyors to respond to production demands automatically, transferring precise quantities on schedule without manual intervention.
Integration and Control Systems
The effectiveness of pharma automation depends heavily on the sophistication of the control systems orchestrating individual components into cohesive workflows. Modern integration approaches connect warehouse management systems, manufacturing execution platforms, and enterprise resource planning software into unified ecosystems where information flows seamlessly and decisions propagate automatically.
These integration layers employ standardised communication protocols, allowing equipment from multiple manufacturers to cooperate within single facilities. Application programming interfaces (API) enable custom integrations where standard protocols prove insufficient, creating connections between legacy systems and modern automation platforms.
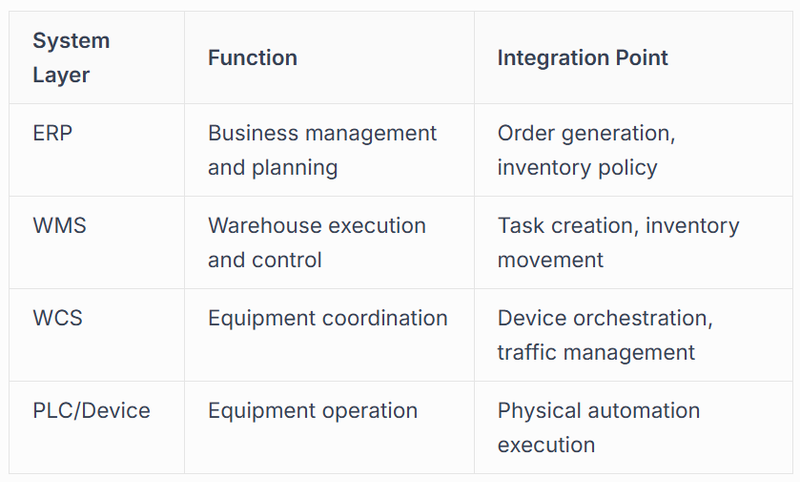

The control hierarchy ensures that business decisions made in ERP systems translate into warehouse actions executed by physical automation. When a customer order enters the system, it flows through these layers, transforming from a commercial transaction into a series of coordinated robot movements, conveyor activations, and verification steps that culminate in accurate, compliant shipment.


Real-Time Visibility and Decision Support
Modern pharma automation systems provide unprecedented visibility into operations, offering real-time dashboards that display inventory positions, system performance, order status, and exception conditions. This visibility empowers managers to make informed decisions quickly, responding to problems before they escalate and identifying opportunities for process improvement.
Predictive analytics built into advanced automation platforms analyse historical performance data to forecast future demands, identify potential equipment failures before they occur, and recommend optimal inventory positioning. These capabilities transform automation systems from reactive tools that execute commands into proactive partners that suggest improvements and anticipate needs.
Scalability and Future-Proofing Pharmaceutical Operations
Pharmaceutical companies must plan automation investments with long-term scalability in mind, selecting technologies and architectures that accommodate growth without requiring complete system replacement. Modular automation approaches allow facilities to start with core capabilities and expand systematically as volumes grow or requirements change.
The goods-to-person systems increasingly popular in pharmaceutical warehousing exemplify this scalability. Facilities can begin with a modest grid serving a single picking station, then expand by adding grid modules and workstations as throughput requirements increase. This incremental growth path spreads capital investment over time whilst maintaining operational continuity during expansion phases.
Future pharmaceutical automation will likely incorporate emerging technologies currently in research and development. Artificial intelligence agents capable of simulating and optimising complex pharmaceutical processes represent one frontier, potentially revolutionising how companies approach drug discovery, formulation, and production planning. Whilst these capabilities remain largely experimental, they illustrate the trajectory of pharmaceutical automation towards increasingly intelligent, autonomous systems.
Preparing for Regulatory Evolution
Regulatory frameworks governing pharmaceutical operations continue to evolve, with authorities worldwide implementing new requirements for serialisation, supply chain security, and quality documentation. Automation systems designed with regulatory compliance at their core adapt more readily to these changes than systems where compliance was an afterthought.
Comprehensive automation platforms incorporating programmable logic controllers (PLC), human-machine interfaces (HMI), and supervisory control and data acquisition (SCADA) systems provide the flexibility to modify processes, update documentation practices, and implement new verification steps as regulations evolve. This adaptability protects automation investments against regulatory obsolescence, ensuring systems remain compliant and valuable across their operational lifespans.
Economic Considerations and Return on Investment
Pharmaceutical automation requires substantial capital investment, making thorough economic analysis essential before committing to major projects. The return on investment (ROI) calculation for pharma automation extends beyond simple labour cost savings to encompass accuracy improvements, compliance cost reductions, and capacity enhancements that enable revenue growth.
Accuracy improvements deliver value through multiple mechanisms. Reduced picking errors eliminate the costs of returns processing, expedited replacement shipments, and potential regulatory actions. Improved inventory accuracy reduces safety stock requirements, freeing capital whilst reducing obsolescence exposure for time-sensitive pharmaceutical products. Enhanced traceability capabilities prevent costly recalls by enabling precise product location and rapid quarantine of affected batches.
Total Cost of Ownership Analysis
Evaluating automation investments requires examining total cost of ownership (TCO) rather than focusing exclusively on initial capital expenditure. TCO analysis incorporates:
- Initial equipment and installation costs
- Integration and commissioning expenses
- Training and change management investments
- Ongoing maintenance and support costs
- Energy consumption and facility modifications
- Software licensing and update fees
These costs must be weighed against tangible benefits including labour cost reductions, accuracy improvements, throughput increases, and space utilisation gains, as well as less quantifiable advantages like regulatory compliance confidence, enhanced customer service, and competitive positioning. The historical evolution of pharmacy automation demonstrates that early adopters of automation technologies often gained sustainable competitive advantages that persisted long after the technology became mainstream.
Workforce Implications and Change Management
Introducing pharma automation into pharmaceutical operations fundamentally changes workforce requirements, shifting emphasis from manual labour towards technical skills, system monitoring, and exception handling. This transformation requires careful change management to ensure workforce acceptance and successful technology adoption.
Rather than simply eliminating positions, thoughtful automation implementations redeploy workers into higher-value roles that leverage uniquely human capabilities. Manual pickers become system operators, monitoring automation performance and handling exceptions that require judgment. Warehouse supervisors evolve into process optimisation specialists, analysing system data to identify improvement opportunities. Maintenance teams expand their skills to encompass robotic systems, vision technology, and integration platforms alongside traditional mechanical and electrical equipment.
Training programmes must begin well before automation systems go live, giving workers time to develop new skills and build confidence with unfamiliar technologies. Hands-on practice with system interfaces, troubleshooting scenarios, and exception handling builds competence whilst reducing anxiety about technology changes. Ongoing training keeps skills current as systems evolve and capabilities expand.
Creating a Culture of Continuous Improvement
Successful pharma automation implementations foster cultures where workers at all levels contribute ideas for system enhancements, process refinements, and operational improvements. Automated systems generate vast quantities of performance data that frontline workers can analyse to identify patterns, spot inefficiencies, and develop solutions. Creating mechanisms for capturing and implementing these insights transforms automation from a static installation into a continuously improving asset.
Regular review sessions where operational teams examine system performance metrics, discuss challenges, and brainstorm improvements keep everyone engaged in optimisation efforts. Recognition programmes that celebrate successful improvement initiatives encourage participation and reinforce the value of worker contributions to operational excellence.
Pharmaceutical operations demand automation solutions that deliver precision, compliance, and scalability whilst handling products requiring careful environmental control and complete traceability. Whether you're managing pharmaceutical distribution, supporting manufacturing operations, or coordinating complex cold chain logistics, intelligent warehouse automation provides the foundation for competitive advantage in an increasingly demanding marketplace. Automate-X specialises in designing and implementing warehouse automation solutions tailored to pharmaceutical and healthcare supply chain requirements, combining proven robotics, sophisticated software platforms, and deep integration expertise to transform your operations whilst maintaining the rigorous standards your products and customers demand.
